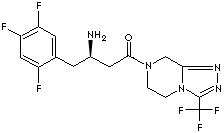
SITAGLIPTIN PHOSPHATE
PRODUCT IDENTIFICATION
654671-77-9 (hydrate)
H.S. CODE
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
Stable under ordinary conditions.
EXTERNAL LINKS & GENERAL DESCRIPTION
Sitagliptin is the first in a new class of once daily, oral anti-hyperglycaemic agents. When added to ongoing metformin or pioglitazone therapy, it significantly improved HbA1c in type-2 diabetic patients who failed to achieve adequate glycaemic control with these agents as monotherapy, and it was found to be non-inferior to glipizide when added to metformin. Sitagliptin treatment was not associated with significantly increased incidences of hypoglycaemia or gastrointestinal adverse events. Sitagliptin as add-on therapy to metformin or a glitazone may be considered as an option for patients who fail to achieve glycaemic control despite an adequate trial of established first and second-line regimens. Comparative and long-term safety and efficacy data are lacking. (http://www.nyrdtc.nhs.uk/)
Sitagliptin (Januvia), the first in a new class of antihyperglycemic agents, was approved in October 2006. It inhibits the enzyme dipeptidyl peptidase-4 (DPP-4), which causes the degradation of glucagon-like peptide-1 (GLP-1). The only other agent on the market that affects levels of GLP-1 is exen atide (Byetta). Exenatide is an injectable GLP-1 agonist, which mimics the action of naturally occurring GLP-1. Indications for sitagliptin include use in type 2 diabetes as monotherapy or in combination with metformin or thiazolidinediones . DPP-4 inhibitors have actions similar to those of the GLP-1 agonist, such as stimulation of insulin secretion, inhibition of glucagon secretion, and preservation of beta-cell function. In contrast, DPP-4 inhibitors do not slow gastric emptying or cause weight loss, possibly due to lower concentrations of GLP-1 postprandially compared to the GLP-1 agonist. DPP-4 inhibitors raise incretin levels to normal or slightly above normal levels. The advantage of DPP-4 inhibitors is oral administration; the GLP-1 receptor agonist is injectable. (http://www.uspharmacist.com/)Dipeptidyl peptidase-IV (DPP-IV) inhibitors are a promising new class of drugs that may potentially play an important role in the management of type 2 diabetes mellitus. These agents improve glycemic control by preventing the rapid inactivation of the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP), leading to stimulation of insulin release from the pancreas, inhibition of glucagon secretion, and potentially an improvement in pancreatic beta-cell function. (http://www.modernmedicine.com/)
Dipeptidyl peptidase-4 inhibitors
|
Product |
CAS RN. |
| Alogliptin |
850649-61-5 |
|
Alogliptin benzoate |
850649-62-6 |
|
Gemigliptin |
911637-19-9 |
|
Linagliptin |
668270-12-0 |
|
Saxagliptin |
361442-04-8 |
|
Saxagliptin hydrate |
945667-22-1 |
|
Sitagliptin |
486460-32-6 |
|
Vildagliptin |
274901-16-5 |
APPEARANCE
PURITY
ISOMER
1.0% max
MELTING POINT
198 - 202 C
LOSS ON DRYING
5.0% max